In many applications with low requirements on visual appearance, the natural oxide offers corrosion protection that is good enough. However, if the aluminium is to be painted or adhesively bonded and used in a corrosive environment, the natural oxide is too poor to ensure the required service life. Pre-treatment is necessary to produce a more stable and well-defined surface.
Let me explain. Aluminium oxide layers can consist of different forms of oxides and hydroxides. The composition depends on the circumstances during formation and the alloying elements and contaminants present. If water is present when the oxide is formed, it can also contain various proportions of crystal water.
The composition affects the stability of the oxide.
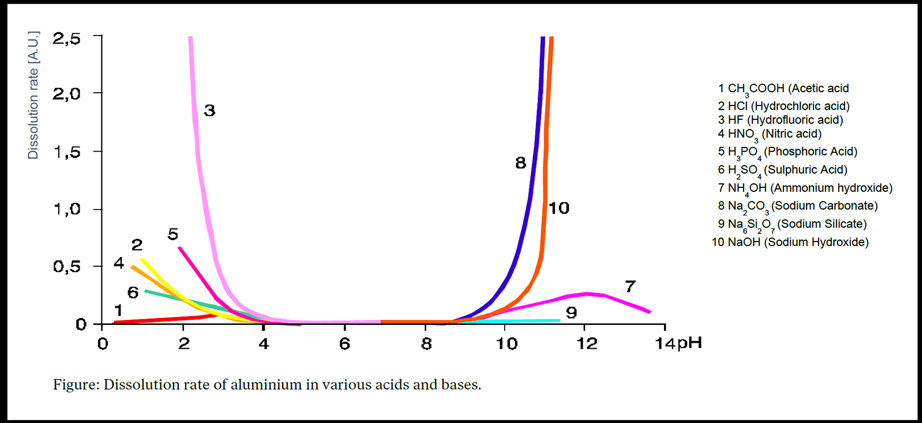
Aluminium oxide is normally stable in the pH range 4 to 9. Below and above these limits, the risk of corrosion attack is much higher. This also means that both acidic and alkaline solutions can be used to etch aluminium surfaces during pre-treatment.
Alloying elements that affect corrosion
Apart from the oxide’s protective properties, the corrosion properties of an aluminium alloy are controlled by the amount of more noble intermetallic particles in the material. If an electrolyte solution such as water or salt is present, corrosion can be initiated and the particles that are more noble than aluminium will act as cathodes while the surrounding areas will become anodes, where the aluminium dissolves.
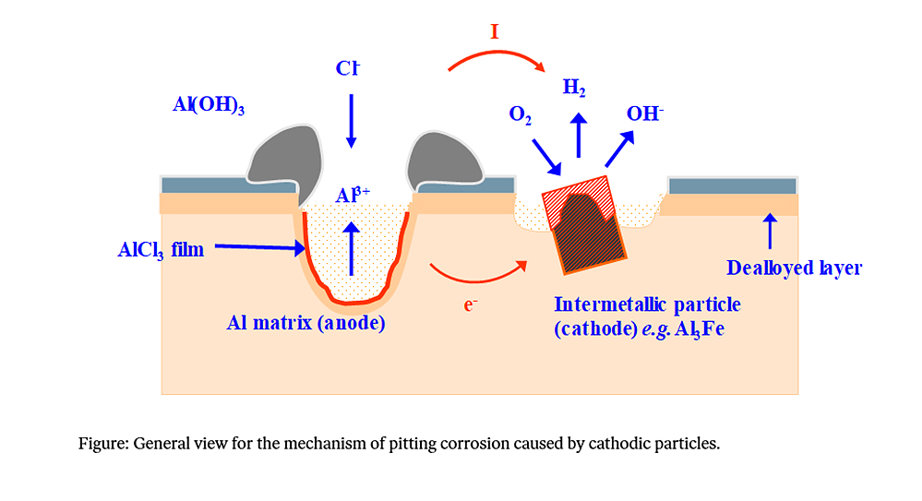
Even particles that only contain small amounts of noble elements may act as pure noble particles because of selective dissolution of aluminium on the particle’s surface. Particles that contain iron cause a significant reduction in corrosion resistance, and copper is also known to reduce corrosion resistance. Higher concentrations of impurities, such as lead, at the grain boundaries, also have a negative effect on corrosion resistance.
Corrosion resistance in 5000 and 6000 series aluminium alloys
Aluminium alloys from the 5000 and 6000 series have relatively low levels of alloying elements and intermetallic particles, which make them relatively resistant to corrosion. In general, high-strength alloys contain larger amounts of alloying elements, which in turn results in reduced resistance to corrosion.
High-strength 2000-series alloys that contain copper (and are used in the aviation industry) often have a thin cladding of pure aluminium to prevent corrosion attack.
Recycled alloys normally have an increased level of trace elements which make them slightly more sensitive to corrosion. But notice that the variation in corrosion resistance between different alloys – and even between materials of the same alloy – with differences in productions method and heat treatment history is often larger than that from trace elements.
My point is that your supplier ought to be able to answer your questions about whether the aluminium product you are considering will meet completely the requirements of your product, especially if corrosion resistance is critical. Get the technical know-how. Because aluminium is not just aluminium.









